Atomic mass of hydrogen8/26/2023 nuclear magnetic resonance spectroscopy. Deuterium and its compounds are used as a non-radioactive label in chemical experiments and in solvents for 1 Water enriched in molecules that include deuterium instead of protium is called heavy water. Presumably the differential concentration of deuterium in the inner solar system is due to the lower volatility of deuterium gas and compounds, enriching deuterium fractions in comets and planets exposed to significant heat from the Sun over billions of years of solar system evolution.ĭeuterium is not radioactive, and does not represent a significant toxicity hazard. Deuterium on Earth has been enriched with respect to its initial concentration in the Big Bang and the outer solar system (about 27 ppm, by atom fraction) and its concentration in older parts of the Milky Way galaxy (about 0.023%, or 23 ppm). Deuterium comprises 0.0026–0.0184% (26 ppm or 184 ppm by population, not by mass) of hydrogen samples on Earth, with the lower number tending to be found in samples of hydrogen gas and the higher enrichment (0.015% or 150 ppm) typical of ocean water. The nucleus of deuterium is called a deuteron. (atomic mass 2.014 101 777 844(15) Da), the other stable hydrogen isotope, is known as deuterium and contains one proton and one neutron in its nucleus. 
(During the early study of radioactivity, some other heavy radioactive isotopes were given names, but such names are rarely used today.)Ī deuterium atom contains one proton, one neutron, and one electron. , with no neutrons, is sometimes called protium. ) instead to avoid confusion in the alphabetic sorting of chemical formulas. 
The IUPAC accepts the D and T symbols, but recommends using standard isotopic symbols ( 2 The symbols D and T are sometimes used for deuterium and tritium. (or hydrogen-2) isotope is deuterium and the 3 Hydrogen is the only element whose isotopes have different names that remain in common use today: the 2 Heavier isotopes also exist, all of which are synthetic and have a half-life of less than one zeptosecond (10 −21 s). Hydrogen ( 1H) has three naturally occurring isotopes, sometimes denoted 1 Therefore, there are various non-equivalent definitions of atomic radius.The three most stable isotopes of hydrogen: protium ( A = 1), deuterium ( A = 2), and tritium ( A = 3). However, this assumes the atom to exhibit a spherical shape, which is only obeyed for atoms in vacuum or free space. The atomic radius of a chemical element is a measure of the distance out to which the electron cloud extends from the nucleus. It must be noted, atoms lack a well-defined outer boundary. The atomic radius of Hydrogen atom is 31pm (covalent radius). Note that, each element may contain more isotopes, therefore this resulting atomic mass is calculated from naturally-occuring isotopes and their abundance. The atomic mass is carried by the atomic nucleus, which occupies only about 10 -12 of the total volume of the atom or less, but it contains all the positive charge and at least 99.95% of the total mass of the atom. The atomic mass or relative isotopic mass refers to the mass of a single particle, and therefore is tied to a certain specific isotope of an element. Mass numbers of typical isotopes of Hydrogen are 1 2. Isotopes are nuclides that have the same atomic number and are therefore the same element, but differ in the number of neutrons. The difference between the neutron number and the atomic number is known as the neutron excess: D = N – Z = A – 2Z.įor stable elements, there is usually a variety of stable isotopes. Neutron number plus atomic number equals atomic mass number: N+Z=A. The total number of neutrons in the nucleus of an atom is called the neutron number of the atom and is given the symbol N. 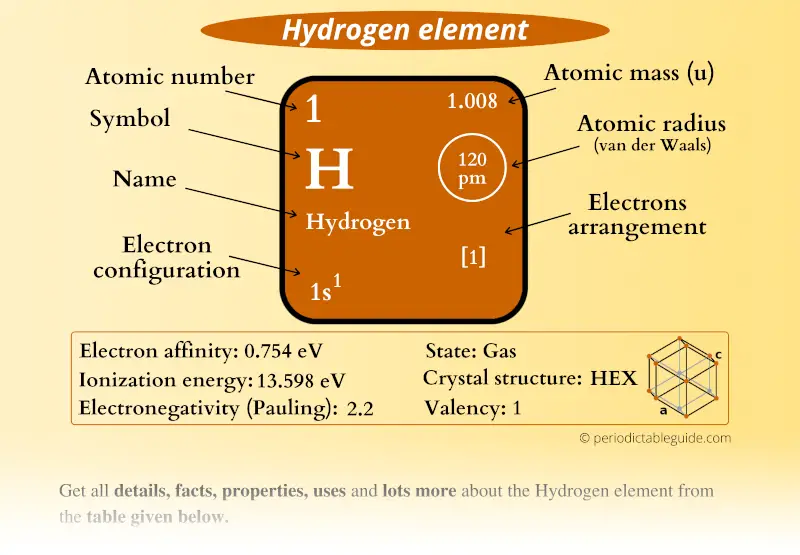
The total electrical charge of the nucleus is therefore +Ze, where e (elementary charge) equals to 1,602 x 10 -19 coulombs. Total number of protons in the nucleus is called the atomic number of the atom and is given the symbol Z. Hydrogen is a chemical element with atomic number 1 which means there are 1 protons in its nucleus. 
Atomic Number – Protons, Electrons and Neutrons in Hydrogen
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
